Electrostatic attraction between oppositive charges is a major force holding interacting biomolecules together in cells. It is traditionally believed that it requires direct contact between biomolecules since their surface is constantly shielded by ions. In a recent study from the Interdisciplinary Research Centre on Biology and Chemistry (IRCBC), Shanghai Institute of Organic Chemistry (SIOC) of the Chinese Academy of Sciences(CAS), scientistsfor the first time discovered that long-range interaction through electric dipole moment (EDM) of proteins controls an important transmembrane signaling pathway. Tumor necrosis factor receptor-1 (TNFR1) is a pro-inflammatory transmembrane receptor with multifaceted functions ranging from inflammation, innate immune to cell death. The outcome of TNFR1 signaling is determined by a complex beneath cell membrane triggered by the engagement of the extracellular ligand. This so-called “Complex-I” is a transient signalosome featuring dynamic assembly and disassembly, which determines the signaling strength of downstream pathways and cell fate. The dynamics of Complex-I is manifested as the rapid recruitment of two intracellular components, TRADD and RIPK1, within a few minutes and the rapid disassociation of RIPK1 after that. Despite the tremendous biological importance of the dynamic nature of Complex-I, the driving force behind the dynamics of Complex-I is a long-standing enigma in receptor biology for decades. 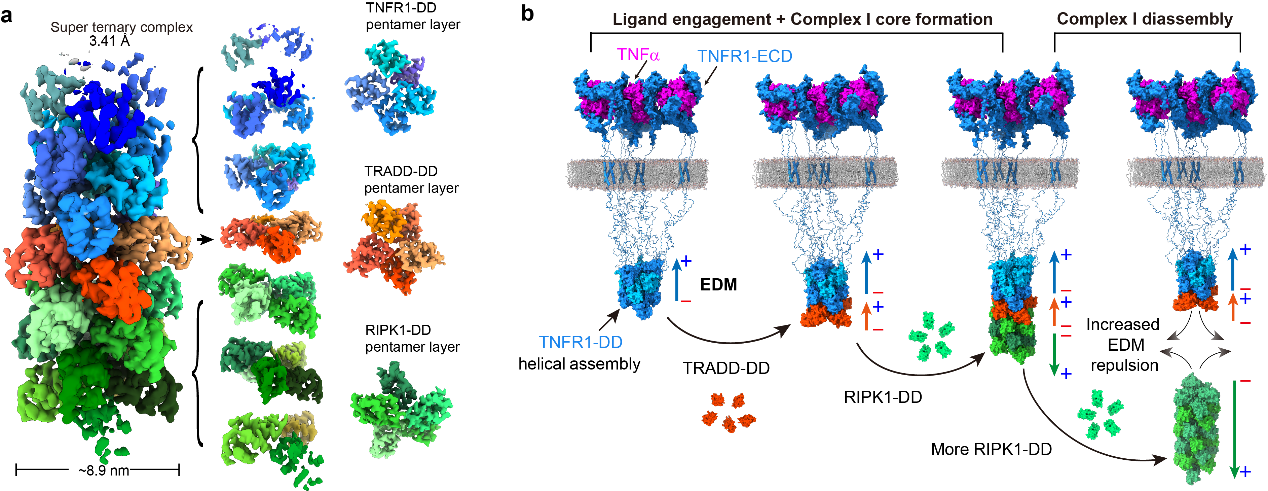
Figure 1. Mechanism for the dynamic assembly and disassembly of TNFR1 signaling Complex-I. (a) A helical super-complex consists of death domains of TNFR1, TRADD and RIPK1. (b) The force between electric dipole moments drives the dynamics of Complex-I. (Image by Jianping LIU) In the study published on Nature with the title “Electric dipole moment drives the dynamics of TNFR1 mediated Complex I signalosome”, scientists from IRCBC captured the core of Complex-I which consists of TNFR1, TRADD and RIPK1. The high-resolution structure determined by cryo-EM technology reveals the structural feature of a helical super-complex assembled by 31 copies of death domains (DDs): a layer of TRADD-DD pentamer is sandwiched between two layers of TNFR1-DD and two layers of RIPK1-DD pentamers. The authors found that the unique mode of assembly is determined by the specific interaction between three types of DDs through contacting surfaces with highly complementary electrostatics. They also noted that the highly polarized distribution of charge on the surfaces of these DDs results in strong electric dipole moment (EDM) in this helical super-complex. Most intriguing is that the direction of EDM of RIPK1 layers is opposed to that of TNFR1 and TRADD layers. In physics, opposing EDMs can cause long-range repulsion between molecules without physical contact. Thus, the scientist proposed that the long-range EDM interaction between RIPK1-DD and TNFR1-DD/TRADD-DD would destabilize DD super-complex and drive the dynamics of Complex-I. With a series of carefully designed mutants of RIPK1 that only change the EDM without affecting DD interacting surface, they proved that increasing the EDM of RIPK1-DD indeed shorten the life-time of RIPK1 in Complex-I, reduce the signaling strength of NF-kB pathway, and vice versa. This work beautifully revealed that EDM interaction between proteins drives the dynamics and function of an important signaling complex, and first time experimentally demonstrated the unprecedented functional role of long-range EDM interaction in cell signaling.
LIU Jianping Ph.D Interdisciplinary Research Center on Biology and Chemistry (IRCBC), Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences Haike Road 100 Shanghai 201204 China Email: jpliu@sioc.ac.cn Tel: +86-21-68582560 |


